While he considers the overall results impressive, he’d like to see a longer follow up, he says, noting that the response to the immunizations was variable. The fact that immune cell counts climbed to the point where researchers could give some of the patients vaccines “is really amazing,” says Charles Venditti, a pediatric geneticist at the National Human Genome Research Institute who was not involved in the study. Gene therapy is going to be the way to go, certainly, for SCID diseases. (The nonresponsive patient was re-treated with the gene therapy, which increased his T cell count.) Four of the patients received vaccinations against tetanus, diphtheria, pertussis, polio, and pneumonia, although only two of those infants mounted a normal immune response to all of the vaccines, as measured by counts of cells and other markers in their blood. The researchers monitored levels of the patients’ immune cells for a median of 16 months all but one of the first eight saw climbing levels of T cells, B cells, and natural killer cells following the treatment. They then treated the babies with one to two doses of busulfan, and injected the gene-edited cells back into them. 
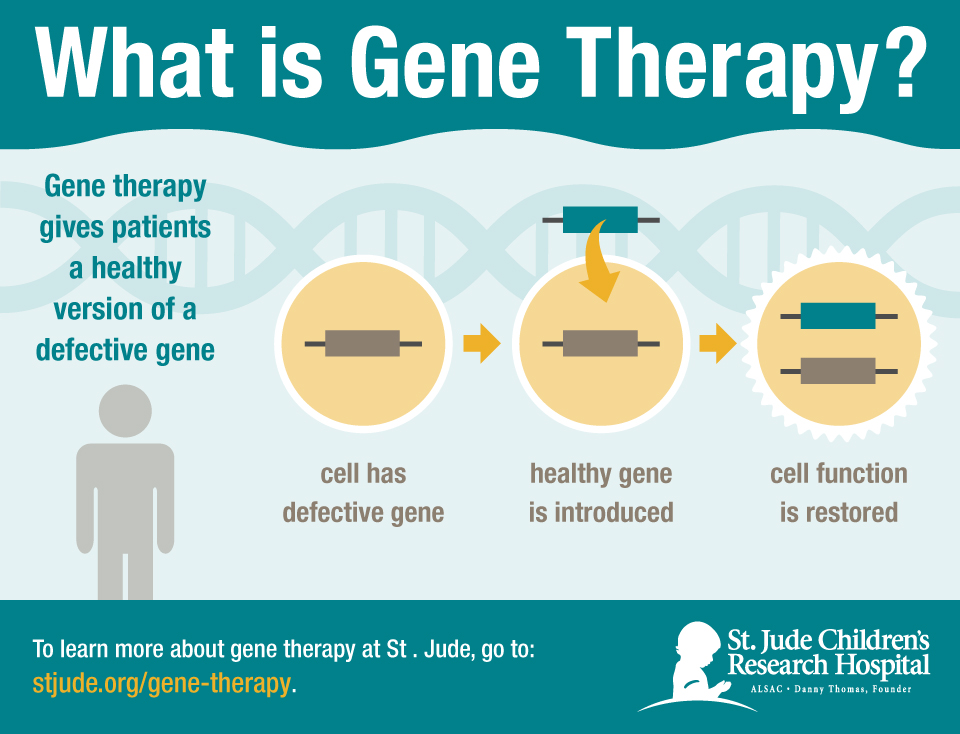
Doctors extracted bone marrow cells from each patient and sent them to a lab at St. They ranged in age from 2–14 months at the time of treatment, and none had a matched sibling donor available. Jude’s (the study reports results for the first eight). Ten infants were treated in the study, half at UCSF and half at St. Its aim was to create space around the bone marrow for the gene-edited cells to get in and take hold. In addition to using a different vector from previous gene therapies, the study protocol called for a novel step, devised by researchers at the University of California, San Francisco (UCSF): pretreatment of patients with a low dose of the chemotherapy drug busulfan, which kills off immune precursors. The IL2RG sequence comes with an insulator sequence at the end designed to prevent it from turning on nearby genes in the area of the genome it lands in, given that switching on the wrong gene could lead to cancer. Jude, delivers a functioning copy of IL2RG into patients’ extracted bone marrow cells using a lentiviral vector modeled on HIV’s shell. The gene therapy in the new study, developed at St. Mamcarz says that even later, safer iterations of gene therapy only restored T cells, not B cells, so patients needed to be treated with immune-boosting immunoglobulins after gene therapy in order to compensate for the missing cells and stay healthy. While they initially produced promising results, the earliest therapies appeared to cause leukemia in some patients. See “ Targeting DNA”Įfforts to treat SCID-X1 with gene therapy began two decades ago. However, only about 20 percent of SCID-X1 patients have a matched sibling, and receiving a transplant from another donor carries a risk of graft-versus-host disease. by far that’s the best case scenario” for current treatment options, says Hey Chong, who heads the Division of Pediatric Allergy and Immunology at Children’s Hospital Pittsburgh and was not involved in the study. The standard treatment for the condition is a transplant of bone marrow tissue that can make normal immune cells, but finding an immunological match for patients can be a challenge. In the most common form of the disease, X-linked SCID (SCID-X1), the gene at fault is IL2RG, which codes for a piece of the cytokine receptors needed for the normal development of several different kinds of immune cells, including T cells, B cells, and natural killer cells. People with severe combined immunodeficiency (SCID) have mutations in genes needed for immune cell function, leaving them vulnerable to infection. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
